 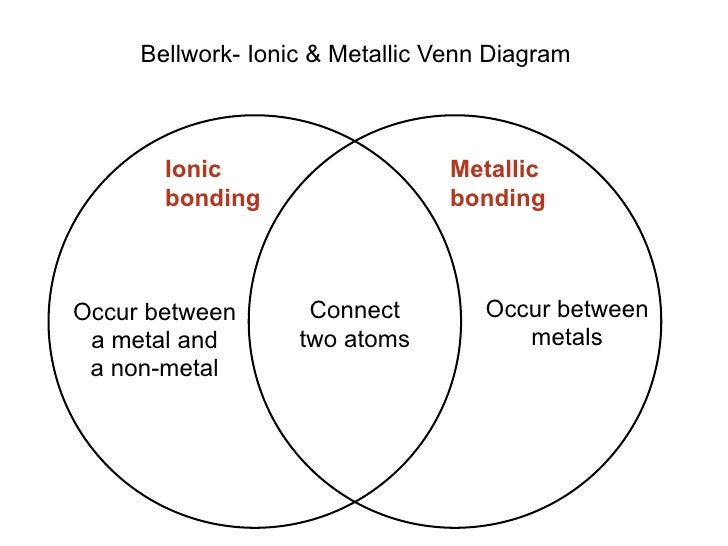
The focus of this section is ionic and covalent bonding. Often, bonding occurs via more than one mechanism. Because of their outstanding versatility, room-temperature ionic liquids (RTILs) are utilized in an ever increasing number of novel and fascinating applications, making them the Holy Grail of modern materials science. Many other types of bonding exist as well. A van der Waals bond occurs due to the attraction of charge-polarized molecules and is considerably weaker than ionic or covalent bonds. The prefix mono- is generally omitted for the first element. A covalent bond occurs when two or more atoms share electrons. 
A complete ionic equation shows all the ions in a solution, while a net ionic equation shows only the ions that participate in a reaction to form products. A molecular equation is valuable because it shows exactly what substances were used in a reaction. Prefixes are used to indicate the number of atoms of a given element in a molecule ( mono- = 1, di- = 2, tri- = 3, tetra - = 4 …). All of these equations have their place in chemistry. While the ions in an ionic compound are strongly attracted to each other, covalent bonds create molecules that can separate from each other when a lower amount of energy is added to them. Name of a binary molecular compound: name of the 1 st element of the formula + name of the 2 nd element with the suffix – ide. Most covalent compounds have relatively low melting points and boiling points. The word ion is omitted. If the metal is a variable charge metal, the charge is indicated in parentheses with Roman numeralsīinary molecular compounds are composed of 2 nonmetals Name of an ionic compound: name of the cation + name of the anion with the suffix – ide for non-polyatomic ions. They consist of 2 ions derived from 2 different elements: one metal element (a cation) and 1 nonmetal element or polyatomic ion (an anion) and CH3OH is interpreted as a molecule with one carbon, four hydrogens. Most ionic compounds are binary compound. MOLECULAR or COVALENT compound, while sodium chloride (table salt) is an IONIC compound. Lewis structures for covalent bonding use lines to represent the sharing of electrons. Write a rule that will allow you to classify a compound as ionic or covalent on. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
